Design Medical Devices for Internet of Things and Your Hardware Capabilities
It seems like everyone I know has a smartwatch or fitness tracker. I gave mine to my mother when I felt like it was making me act weird (I sometimes put my laundry away one item at a time instead of using a laundry basket to get those extra step counts). There are days when I still covet graphs informing me of my status sleeping, walking, and eating.
Fitness trackers barely scratch the surface of capabilities for medical IoT, though. Medical IoT is doing more than helping you optimize your day-to-day life, though. It is being used for everything from helping people manage chronic conditions to improving the hospitals manage patient care.
How is medical IoT used?
The form factor and design of medical IoT devices depends on how and where they are being used. For example, in a hospital, integrating internet connectivity into existing hardware, like an ECG monitor, requires adjustments to an existing design. Novel applications, such as patient location tracking, may require a totally new design or integration into something being used for another purpose like a patient wrist bracelet.
Wearables are at the opposite end of the spectrum from hospital hardware. Devices must integrate into users’ daily lives fairly unobtrusively. A approach is a bracelet or a watch since these are items that people are used to wearing.
A watch can collect a lot of sensor information from the body and environment, but, dependent on data, may not be an ideal location. ECG data is noisy on the limbs and not all sensors function well on the arm, like measuring sun exposure or temperature.
Medical IoT can track patient conditions and alert doctors or nurses when something changes.
Home-based health care falls somewhere in the middle. While many wearables are “fitness trackers” or receive similar labeling, some are more strictly medical devices that require much more rigorous testing and certification. Products like insulin monitors, seizure tracking, or fall identification existed before IoT, but have been modified for connectivity to provide alerts and data tracking.
What are Hardware Considerations?
When designing for medical IoT, the hardware is even more of a challenge than regular medical device design. Obviously, the sensing and monitoring capability is central to the function of your product, and fortunately, there is a huge range of sensors available:
- Accelerometers can track motion during sleep, steps, falls, or other activities.
- ECG tracking is made possible with analog front end systems
- Ultrasound for imaging systems is becoming more compact and easier to deploy
- Sensor ICs abound for everything from biochemistry to temperature.
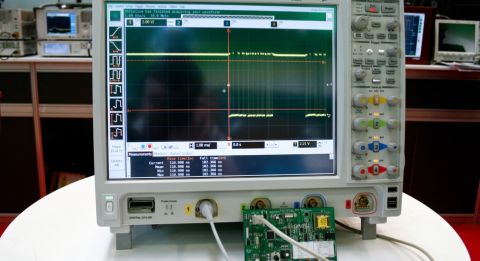
Another major consideration of your design is the microcontroller. The amount of on-board processing you need to do determines how much capability you need. There will probably be some signal processing from your sensors, output to the user, and managing data storage and tracking is necessary before uploading data to the cloud, or a base station.
One of the greatest challenges that sets IoT apart from the previous generation of medical devices is power management. Not only are the issues of electrostatic discharge, and sensitive components increased because of the complex operating environment, but you have to manage everything with a reduced power budget. Remember, these devices, especially the wearables, have to be small enough to be transportable. That means you don’t have wall power, and can’t count on large (and heavy) batteries in your designs.
You don’t want to add weight to an insulin pump that someone needs to wear every day.
When you design medical products for the internet of things, there are numerous requirements to get certified as an actual medical product. You can make design easier by incorporating those requirements into your PCB design software to help you select and manage safety margins from the very beginning.
With in-depth design rule checking incorporated into their unified design environment methodology, you can transfer between checking that all your required ducks are in their respective rows and your design layout with ease in Altium Designer®.
If you’re interested in learning more about how to optimize your PCB design software for the specific design limitations that you encounter, consider talking to an expert at Altium today.